Shared Investigator Platform
Login, e-sign, and manage cases
Cognizant Technology Solutions
iPhone Screenshots






iPad Screenshots





Description
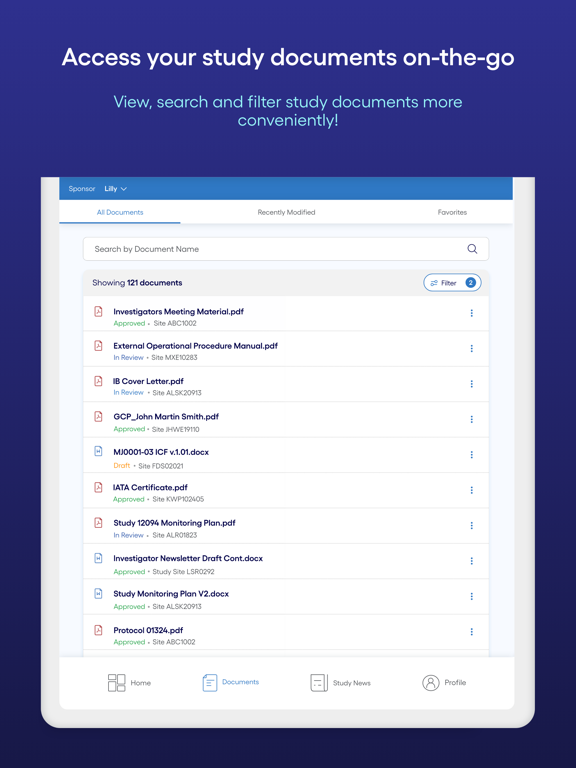
Manage pending actions and accept study invitations. View and generate FDA Form 1572. Access study news and acknowledgments. Log in with existing credentials. Enable biometric authentication for login and E-signature.
The Shared Investigator Platform (Cognizant SIP) Mobile App features a user friendly interface to navigate and manage your actions on the Shared Investigator Platform. With this app, site users can easily login and E-sign using their existing SIP credentials and enable biometric authentication to simplify the subsequent login and E-signature processes. This app offers an easy review of the pending actions, accept study invitations, View and generate FDA Form1572, view the study news, acknowledge safety notifications, view study documents, complete Document Exchange tasks published by your sponsor organizations and receive push notifications of latest study news published and assigned tasks.
App information from Apple App Store. Shared Investigator Platform and related trademarks belong to Cognizant Technology Solutions.